The U.S. Food and Drug Administration (FDA) doesn’t stop monitoring drugs once they’re on the market. In fact, that’s when the real safety work begins. Since January 2025, the FDA has issued at least eight major Drug Safety Communications - each one a red flag for doctors, pharmacists, and patients. These aren’t routine updates. They’re urgent, data-driven alerts that change how medications are prescribed, used, and even withdrawn from shelves. If you or someone you know takes prescription drugs, especially for chronic pain, ADHD, Alzheimer’s, or mental health, you need to know what’s changed.
What Exactly Is a Drug Safety Communication?
A Drug Safety Communication (DSC) is the FDA’s official way of telling the public: something we didn’t see in clinical trials is now showing up in real-world use. These aren’t rumors. They’re based on millions of real patient reports, electronic health records, and post-market studies. The FDA uses Section 505(o)(3) of the Federal Food, Drug, and Cosmetic Act to force drugmakers to update labels, add warnings, or even remove safety programs. These communications are legally binding - manufacturers must comply.
For example, in July 2025, the FDA required every opioid painkiller in the U.S. to carry new labeling. For the first time, they had to include quantitative risk numbers: 1 in 12 patients who take opioids for more than 90 days will develop an opioid use disorder. That’s not vague. That’s a hard number, pulled from two massive post-market studies involving all major opioid manufacturers. This change affects 46 different products and billions in sales. It’s not about scaring people - it’s about giving doctors facts they never had before.
Key Alerts from 2025: What Changed
Here are the most urgent safety updates you won’t find on your pharmacy’s website:
- Opioids (July 31, 2025): All extended-release and long-acting opioids now carry warnings about long-term addiction risk, overdose potential, and new dangers like toxic leukoencephalopathy - a rare brain injury linked to opioid overdose that doesn’t always look like a typical overdose. The FDA also updated drug interaction warnings to include gabapentinoids (like Neurontin and Lyrica), which when mixed with opioids, increase overdose risk.
- ADHD Stimulants (June 30, 2025): Extended-release methylphenidate and amphetamine products (like Adderall XR, Concerta, Vyvanse) now require weight monitoring in children under 6. These drugs can suppress appetite so severely that growth delays occur. Doctors must check weight at baseline and every 3 months.
- Antihistamines (May 16, 2025): Zyrtec (cetirizine) and Xyzal (levocetirizine) now carry a warning about increased risk of seizures in patients with a history of epilepsy or seizure disorders. This was based on over 1,200 case reports submitted to the FDA’s adverse event database.
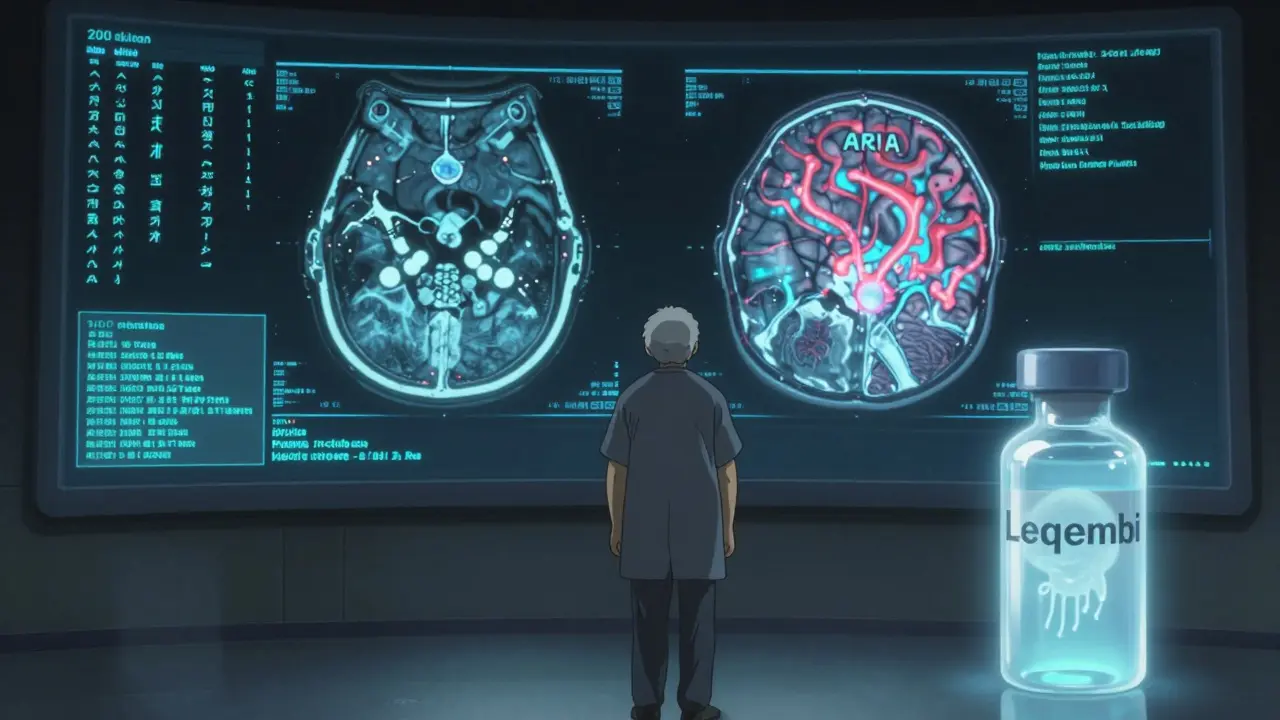
- Alzheimer’s Drug (August 28, 2025): Leqembi (lecanemab) users now need two MRIs - one at 5 months and another at 14 months after starting treatment. This is because of amyloid-related imaging abnormalities (ARIA), which occurred in 274 patients during the first year of use. These brain changes can cause swelling or bleeding. The FDA didn’t pull the drug - it just said: monitor closely.
- mRNA Vaccines (June 25, 2025): Comirnaty (Pfizer) and Spikevax (Moderna) now include updated data on myocarditis and pericarditis. The FDA confirmed 1,195 cases per million second doses in males aged 12-29. That’s rare, but real. The warning now includes age-specific risk levels and guidance on when to seek care.
Not all changes are warnings. In June 2025, the FDA removed the Risk Evaluation and Mitigation Strategy (REMS) for clozapine - an antipsychotic used for treatment-resistant schizophrenia. Why? Because after 35 years of use and millions of doses, the data showed that routine blood monitoring for agranulocytosis (a rare blood disorder) was no longer needed for most patients. This was a rare win: less bureaucracy, more trust in clinical judgment.
Who’s Affected? Real Numbers Behind the Alerts
These aren’t theoretical risks. Millions of people are directly impacted:
- Over 9.4 million U.S. children and teens take ADHD stimulants - many on long-term therapy.
- 25 million Americans use cetirizine or levocetirizine for allergies.
- More than 11 million adults are prescribed opioids for chronic pain.
- Over 300,000 people with Alzheimer’s are on Leqembi or similar drugs.
The opioid labeling update alone affects a market worth $11.3 billion in 2024. But here’s the twist: 87% of those prescriptions are for generic versions. That means even if you’re taking a cheap, off-brand painkiller, the new warning applies to you.

What Should You Do? Practical Steps
If you’re on any of these medications, here’s what to do - no panic, just action:
- Check your prescription. Does it match the drug names listed above? If yes, don’t stop cold turkey. Talk to your doctor.
- Ask about monitoring. If you’re on Leqembi, ask if you’ve had your MRIs. If you’re on ADHD meds and your child is under 6, ask about weight tracking. If you’re on opioids, ask if your pain plan still makes sense after 90 days.
- Review your meds list. Gabapentin and opioids together? That’s now flagged as a high-risk combo. Talk to your pharmacist.
- Use the FDA’s Medication Guides. They’re free, available in 18 languages, and updated with every DSC. Find them at fda.gov/drugsafety.
Many patients feel confused or scared. That’s normal. But the goal isn’t to scare you - it’s to empower you. The FDA doesn’t issue these alerts lightly. They’re based on hard data, not fear.
Why This Matters: The Bigger Picture
The number of Drug Safety Communications has jumped 44.7% since 2020. Why? Because we’re using drugs longer, more often, and in combination. A 70-year-old with arthritis might take an opioid, a statin, a blood thinner, and a sleep aid - all at once. We didn’t have enough data on how these mix. Now we do.
The FDA’s Sentinel Initiative - which tracks health data from 300 million patients across 25 U.S. health systems - is now detecting signals faster than ever. By 2027, it’s expected to find 2-3 times more safety issues than it did in 2024. That means more alerts, more updates, and more changes ahead.
Pharmaceutical companies are responding. Post-market safety studies are now costing 28.5% more than they did in 2020. The opioid consortium alone spent $187 million on the studies that led to the July 2025 labeling update. This isn’t just bureaucracy - it’s science catching up with real-world use.
What’s Next?
The FDA’s 2026-2030 plan says this: all drugs with black box warnings will soon be required to generate real-world evidence. That means if a drug has the strongest possible warning, the manufacturer must prove it’s still safe in everyday use - not just in controlled trials. This could change how drugs are developed and priced.
Expect more frequent alerts. The FDA now aims to issue safety communications within 30 days of confirming a risk - down from 60-90 days. That’s a big deal. It means you’ll hear about dangers faster.
But here’s the catch: doctors are overwhelmed. A survey by the American Medical Association found that 63% of primary care doctors feel they don’t have enough time in a 15-minute visit to explain these new risks. And 41% say they don’t have access to good non-opioid pain options. That’s the real challenge - not the science. It’s the system.
Final Thoughts
Medication safety isn’t about perfection. It’s about awareness. The FDA isn’t saying stop taking your pills. It’s saying: know the risks. Monitor. Ask questions. Whether you’re managing chronic pain, treating ADHD, or caring for someone with Alzheimer’s, these updates are meant to help you make better decisions - not worse ones.
Stay informed. Talk to your provider. Don’t ignore a label change because it’s inconvenient. The data is out there. And now, so are the answers.
Are drug recalls the same as Drug Safety Communications?
No. A drug recall means the product is pulled from the market because it’s unsafe, contaminated, or mislabeled. A Drug Safety Communication is a warning - it doesn’t mean the drug is taken off shelves. Most DSCs ask doctors and patients to monitor more closely, adjust dosages, or check for new side effects. Only in rare cases does a DSC lead to a recall.
I’m on an opioid for chronic pain. Should I stop?
No. Stopping suddenly can cause dangerous withdrawal symptoms. Instead, schedule a conversation with your doctor. Ask: "Is this still the best option for me?" and "Have we considered non-opioid alternatives?" The new labeling doesn’t mean you’re at risk - it means you should have an honest discussion about long-term use.
Can I trust the FDA’s safety alerts?
Yes. The FDA doesn’t issue these alerts based on anecdotal reports. Each one is backed by real data from millions of patient records, clinical studies, and post-market surveillance. They’re reviewed by independent advisory committees. While no system is perfect, the FDA’s process is the most rigorous in the world. If you’re unsure, ask your pharmacist or doctor to show you the official communication on fda.gov.
I take Zyrtec. Should I be worried about seizures?
Only if you have a history of seizures or epilepsy. For most people, Zyrtec is safe. The new warning applies to a small group - those with pre-existing seizure disorders. If you’ve never had a seizure, there’s no need to stop. But if you’re unsure, talk to your doctor. They can review your medical history and decide if it’s still right for you.
Where can I find the official Drug Safety Communications?
Go directly to the FDA’s Drug Safety Communications page: fda.gov/drugsafety. All communications are archived by date and drug name. You can search by medication, date, or keyword. This is the only source that’s legally authoritative.





phyllis bourassa
I just got off the phone with my pharmacist and she said the new opioid labeling is a total game-changer. Like, I’ve been on oxycodone for 7 years and no one ever told me 1 in 12 people get addicted. 1 in 12! That’s not a statistic, that’s a ticking time bomb. I’m scheduling a chat with my doctor tomorrow. No more pretending this is just ‘a little help’.
Also, gabapentin + opioids? Yeah, I take both. Guess I’m lucky I didn’t OD while napping on the couch watching Netflix.
Pranay Roy
This whole FDA thing is a scam. They’re pushing this because Big Pharma paid them to scare people into switching to cheaper generics. Did you know the FDA director’s wife works for a generic drug company? The real story? They want you off branded meds so they can control the market. Leqembi? It’s fine. The MRIs? Just a way to make hospitals rich. They’re not protecting you - they’re protecting profits.
Joe Prism
The system’s not broken. It’s evolving.
We used to treat drugs like magic bullets. Now we’re learning they’re more like fireworks - beautiful until they go sideways. The FDA’s finally catching up. Not perfect. Not fast enough. But moving in the right direction.
Bridget Verwey
Oh wow. So now we’re supposed to be ‘empowered’ by a 12-page PDF no one reads? Let me guess - the same people who didn’t read the warning labels on their supplements are now going to read the FDA’s 47-point DSC on Leqembi? 😂
Meanwhile, my 82-year-old mom is still taking 11 pills a day and thinks ‘ARIA’ is a new yoga pose. This isn’t empowerment. It’s bureaucratic noise.
Andrew Poulin
Stop overthinking it. If you’re on an opioid longer than 90 days you’re already in trouble. Period. The FDA didn’t create the problem. You did. Go talk to your doctor. Or don’t. But don’t act surprised when your brain turns to mush.
Weston Potgieter
So Zyrtec causes seizures? Cool. That’s the same stuff my aunt takes for her ‘seasonal sneezes’ while she’s on 3 other meds and chugging red bull. Meanwhile, I’m on Adderall, Lexapro, and a muscle relaxer. I’m basically a walking pharmacy. If the FDA’s gonna start warning me about every combo I’ve got, they better start printing warnings on my coffee mug too.
Also - why’s no one talking about how the REMS removal for clozapine was the only good thing here? That’s the real win. The rest is just noise.
Vikas Verma
The integration of real-world evidence into regulatory frameworks represents a paradigm shift in pharmacovigilance. The Sentinel Initiative leverages distributed data networks to achieve unprecedented signal detection velocity. This operational transformation enables proactive risk mitigation rather than reactive containment. The statistical significance of the opioid use disorder incidence rate (8.33%) confirms the necessity of quantitative labeling under Section 505(o)(3).
Sean Callahan
i just took my mom’s zyrtec yesterday because my nose was stuffy and now i’m paranoid i’m gonna have a seizure lmao
also my dad’s on opioids and he says he feels fine so why are they making such a big deal? like... is this even real? or is this just fearmongering? idk anymore
Ferdinand Aton
You know what’s wild? The FDA didn’t warn about gabapentinoids with opioids until 2025. But in 2019, a guy in Nebraska died from mixing them. His obituary said he was ‘peaceful in his sleep.’ Coincidence? I think not.
William Minks
This is why I love science 🤝🧠
Real data > anecdotes. Real monitoring > guesswork. The FDA’s doing the hard work so we don’t have to. Grateful for the transparency. Keep it coming.
Jeff Mirisola
I’ve been on Vyvanse since I was 14. Now I’m 32. My doctor never checked my weight. I lost 20 lbs in a year and thought I was ‘just getting fit.’ Turns out, I was just starving. This alert? Lifesaver. I’m getting my kid’s weight checked next week. No more winging it.
Susan Purney Mark
I’m a nurse. I’ve seen too many patients get scared and quit their meds cold turkey. Please don’t do that. If you’re on Leqembi or opioids or ADHD meds - talk to your provider. Not your Reddit buddy. Not your cousin who ‘read an article.’ Talk to someone who’s seen your chart.
And if you’re scared? I got you. DM me. I’m here.
Ian Kiplagat
Interesting. The UK’s MHRA issued a similar alert on cetirizine last year. We’ve had 17 reported cases. None fatal. Still - better safe than sorry. I’ve switched to loratadine. Simple swap. No drama.
Amina Aminkhuslen
Let’s be real - if the FDA had warned about Zyrtec in 2018, my sister wouldn’t have had a seizure in the middle of her yoga class. She’s 24. No history. Just ‘allergy meds.’ Now she’s on Keppra. I’m not mad. I’m just... disappointed. This was preventable.
Adebayo Muhammad
The FDA, as a regulatory body, is inherently compromised by its structural dependence on pharmaceutical industry funding, as evidenced by the revolving door between corporate lobbying and regulatory appointments. The 2025 opioid labeling update, while ostensibly data-driven, fails to address the root cause: the commodification of pain management in a neoliberal healthcare system. The real crisis is not pharmacological, but epistemological - we have been trained to trust institutions that profit from our suffering.